Excelvision Eye Drop Recalls
FDA has requested companies whose eye drops were made at Excelvision (a contract manufacturer in France) to recall unexpired lots of eye drops produced at this facility due to adverse inspection findings.
Products affected
Recalls have been announced for the following products:
Genteal Tears Lubricant Eye Gel
All unexpired lots
This recall applies ONLY to the gel - not to Genteal Ointment or to any other Genteal brand eye drops
Systane Night Gel
All unexpired lots
This recall applies ONLY to the gel - not to Systane Ointment or to any other Systane brand eye drops
Optase Dry Eye Intense (multi-dose preservative-free bottle version only)
This applies only to Optase Dry Eye Intense multi-dose preservative-free bottles only. It does not applies to Optase DEI in single-use vials or to any other Optase brand eyey drop.
Recalled lots:
Lots 8T98, 9T31, 9T32 expiring April 30, 2026
Lots 2V13, 2V14, 2V15 expiring June 30, 2026
Lots 3V35 expiring August 31, 2026
Lots 3V36, 3V37 expiring September 30, 2026
Lots 5V45, 5V46, 9V12 expiring March 21, 2027
Lots 1X57, 1X70, 1X84 expiring May 31, 2027
Recalls are expected for the following products:
Oasis Tears (multi-dose preservative-free bottle version only)
Only 3 lots, expiring August 2026 or earlier
Recall does not apply to Oasis Tears single use vials or to any other Oasis brand eye drops. All Oasis brand eye drops are made at a different facility now.
At least 3 more brands are understood to have been asked by the FDA to conduct recalls of products
Why are these eye drops being recalled?
All of these eye drops were or are manufactured by Excelvision, a contract manufacturing facility in France.
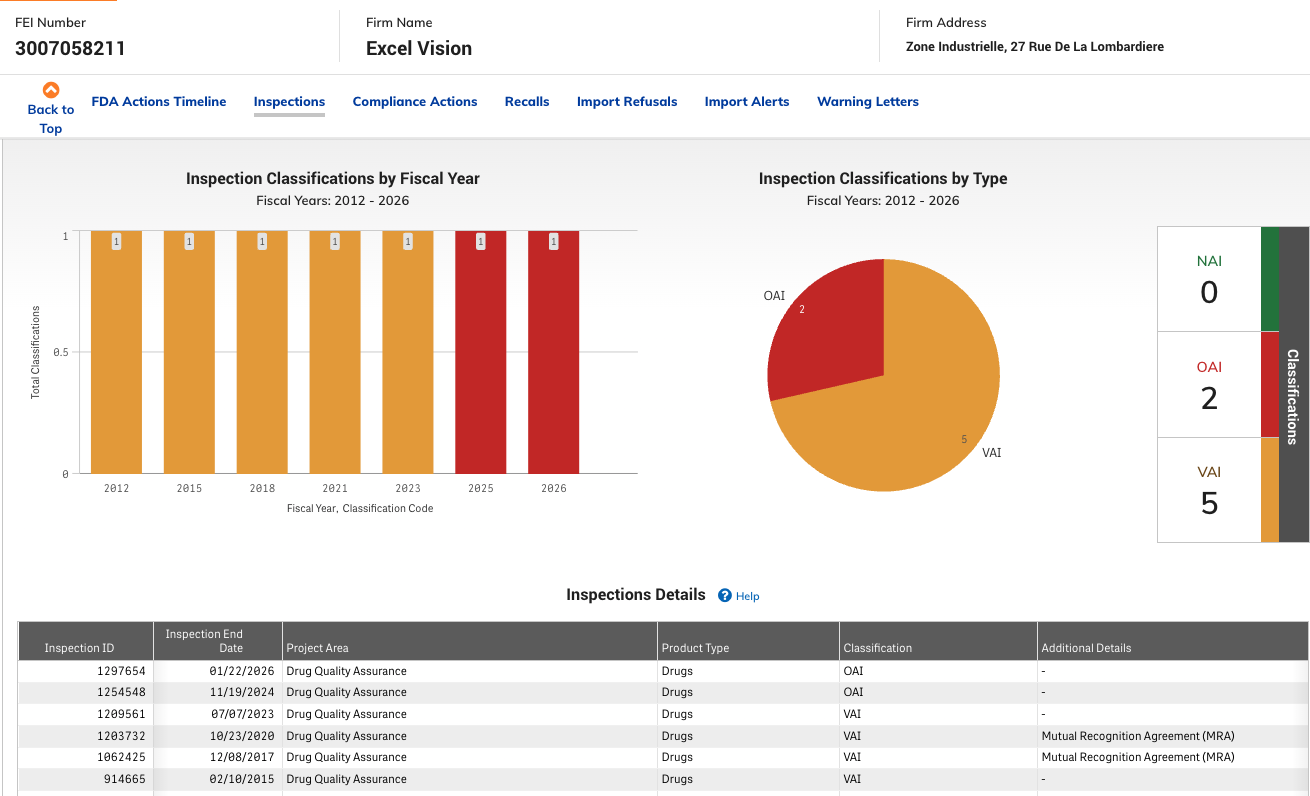
FDA inspections in 2024 and 2025 revealed manufacturing violations that may affect product quality.
Who is Excelvision?
Excelvision is a contract manufacturing facility in France which makes (or made) several name-brand eye drops sold in the US.
In the past two years, FDA has taken enforcement actions against Excel for manufacturing violations. These violations include failing to follow procedures designed to prevent microbiological contamination and failing to properly investigate customer complaints.
Timeline:
November 2024: FDA inspects Excelvision and identifies serious manufacturing violations.
2024 to 2025: Some companies transfer their eye drops to other manufacturing facilities
May 2025: FDA publishes its official Warning Letter, making details of Excel's violations public. Like most FDA Warning Letters to manufacturers, this letter was sent after the company was given the opportunity to correct the problems and their response was deemed inadequate.
January 2026: FDA re-inspects Excelvision resulting in 11 citations for procedural violations, 6 of which were related to aseptic processing and prevention of microbiological contamination.
April 2026: FDA has reportedly asked 6 companies distributing eye drops in the US to recall eye drops manufactured by Excel "to the retail level", meaning consumers do not have to be notified.